The FDA treats pet food packaging with the same safety standards as human food packaging. This means strict regulations under the Federal Food, Drug, and Cosmetic Act (FD&C Act) apply. Here's what you need to know:
- Packaging Safety: Materials that touch pet food are classified as Food Contact Substances (FCS) and must meet FDA safety standards.
- Labeling Rules: Pet food labels must provide clear, accurate details like product name, intended species, ingredients, and feeding directions.
- Compliance Consequences: Non-compliance can lead to recalls, penalties, or even legal action.
-
2026 Updates:
- Mandatory GRAS Submissions: Companies must now notify the FDA about safe packaging materials.
- BHA Reassessment: The FDA is reviewing the safety of BHA, a common preservative.
- Traceability Requirements: FSMA Rule 204 deadlines have been extended, but traceability remains a focus.
Staying compliant isn't just about avoiding penalties - it also protects your brand and ensures pet food safety.
Material Safety and Food Contact Substances (FCS)
The FDA classifies any material that comes into contact with pet food as a Food Contact Substance (FCS). These substances must demonstrate a reasonable certainty of no harm under their intended use. This evaluation considers their migration potential, cumulative dietary exposure, and safe exposure limits . The FDA determines acceptable migration levels and establishes safe exposure thresholds.
Food Contact Substance Regulations Explained
The regulatory status of packaging materials depends on the compliance of each individual substance they contain. For substances expected to migrate into food, compliance can be achieved through one of five pathways:
- Listing as a regulated indirect additive in 21 CFR Parts 174–179
- GRAS (Generally Recognized as Safe) status
- A prior sanction letter
- A Threshold of Regulation (TOR) exemption
- An effective Food Contact Notification (FCN)
The FCN process includes a required 120-day review period . If no objections arise during this time, the notification becomes effective. However, FCNs are manufacturer-specific and apply only to the particular substance and conditions of use outlined in the notification. The FDA clarifies:
"An FCN is effective for the manufacturer, the Food Contact Substance (FCS), and the conditions of use identified in the notification and not effective for a similar or identical substance produced or prepared by a manufacturer other than a manufacturer identified in the prior notification." [6]
For substances with extremely low migration levels (0.5 parts per billion or less), manufacturers may request a TOR exemption. This assumes that such minimal dietary exposures are negligible. To test migration potential, the FDA recommends using food simulants - like 10% ethanol for aqueous foods or corn oil for fatty foods - under accelerated conditions, such as 104°F (40°C) for 10 days, mimicking long-term storage. Packaging manufacturers are also expected to provide a "letter of guaranty" certifying compliance with applicable regulations. Pet food companies should obtain this letter as part of their verification process. Additionally, consulting the FDA's public resources, such as the Inventory of Effective Food Contact Substance Notifications and the Indirect Additive Database, can help confirm the regulatory status of packaging materials.
These pathways provide a framework for understanding how GRAS status applies to packaging materials.
GRAS Status and Packaging Materials
GRAS status offers another route for packaging materials to meet regulatory requirements. Substances with GRAS designation are excluded from the definition of "food additive", meaning they do not require FDA premarket approval. GRAS status can be established either through scientific procedures or a history of common use in food. However, safety assessments must address the needs of the target animal - and, when relevant, the safety of human consumers for products derived from animals.
The FDA explains:
- "Any substance that is intentionally added to food is a food additive, that is subject to premarket review and approval by FDA, unless the substance is generally recognized, among qualified experts, as having been adequately shown to be safe under the conditions of its intended use." [11]
To verify if a packaging component has GRAS status, pet food companies can refer to 21 CFR Parts 182, 184, and 186 or check the FDA's Animal Food GRAS Notices Inventory. Even GRAS substances require evaluation for migration levels that could pose safety risks. For example, in February 2025, the FDA issued a safety communication regarding Areca catechu dinnerware, emphasizing that GRAS status must be re-evaluated when new data suggests potential risks. This highlights that GRAS status is not a permanent designation - the FDA can reassess substances if emerging evidence suggests they no longer meet safety standards.
sbb-itb-0c3a5ed
Labeling Requirements for Pet Food Packaging
The FDA, under 21 CFR Part 501, requires pet food labels to provide clear and accurate product details without making misleading therapeutic claims. This section breaks down the essential elements of pet food labeling and highlights claims that are not allowed. Just like safe packaging materials, accurate labeling plays a key role in maintaining the integrity of the pet food supply chain.
Required Label Elements
Pet food labels must include eight specific elements, split between two main areas:
- Principal Display Panel (PDP): This is where you'll find the brand name, product name, the intended species (like "dog food" or "cat treats"), and the net quantity statement, which must appear in both pounds/ounces and metric units.
- Information Panel: This section includes the guaranteed analysis, a list of ingredients (ordered by weight), a nutritional adequacy statement, feeding directions, and the contact information for the manufacturer or distributor.
The text on labels must be at least 1/16 inch in height, or 1/32 inch for packages smaller than 12 square inches. Additionally, the net quantity statement must be placed within the bottom 30% of the PDP to meet federal Uniform Packaging and Labeling regulations. According to 21 CFR § 501.1, the PDP must be:
"large enough to accommodate all the mandatory label information required to be placed thereon by this part with clarity and conspicuousness and without obscuring design, vignettes, or crowding" [4].
Prohibited Claims and Misleading InformationClaims that suggest a food can treat diseases or modify body functions classify the product as a "new animal drug", which would require separate FDA approval [3][1]. For example, phrases like "cures cancer", "reduces inflammation", or "treats arthritis" are not allowed on pet food labels.
The term "veterinarian approved" is also prohibited. On the other hand, "veterinarian recommended" can be used, but only if supported by a survey involving at least 300 veterinarians. AAFCO clarifies:
"Veterinarians do not approve labels or products, only state regulatory agencies can do that" [12].
Misleading claims can lead to regulatory penalties. For example, the term "dietary supplement" cannot be used, as the Dietary Supplement Health and Education Act applies only to human food. Additionally, highlighting a brand name or ingredient in a way that implies a nutritional benefit or ingredient inclusion that doesn’t exist is considered deceptive.
Specific naming rules are in place to ensure clarity. For instance, "Beef Dog Food" must contain at least 95% beef (excluding water), "Beef Dinner" must have at least 25%, and "Dog Food with Beef" only requires 3% beef. Marketing terms like "new" or "improved" are restricted to a six-month window after a product's launch.
2026 Regulatory Updates Affecting Packaging

2026 Pet Food Packaging Regulatory Updates: GRAS, BHA, and FSMA Rule 204
In 2026, the FDA transitioned pet food packaging oversight from a voluntary system to a mandatory framework, prompting manufacturers to revise their processes and documentation. Key areas impacted include GRAS submissions, chemical reviews, and traceability protocols.
Mandatory GRAS Submissions for Packaging Materials
The FDA is set to overhaul the GRAS (Generally Recognized as Safe) process for packaging materials, proposing a rule in spring 2026 to remove the "self-affirmed" GRAS system. Previously, companies could internally determine that a substance was GRAS without notifying the FDA. Under the proposed rule, companies must inform the FDA of any substances they believe are safe before use, including those indirectly introduced through food packaging. Substances already listed as GRAS or with a "no questions" letter are excluded from this requirement. Manufacturers should consult the FDA's GRAS notice inventory to confirm if their materials are pre-approved or if new submissions, including safety data and technical specifications, are necessary.
BHA Reassessment and Packaging Implications
On February 10, 2026, the FDA initiated a reassessment of BHA, a widely used antioxidant in pet food and packaging. Originally classified as GRAS in 1958 and approved as a food additive in 1961, BHA is now under scrutiny as part of the FDA's Human Foods Program 2026 Priority Deliverables. The National Toxicology Program has flagged BHA as "reasonably anticipated to be a human carcinogen". Addressing this, Health and Human Services Secretary Robert F. Kennedy, Jr. remarked, "This reassessment marks the end of the 'trust us' era in food safety. If BHA cannot meet today's gold-standard science for its current uses, we will remove it from the food supply". Should the FDA find BHA unsafe, it may be removed from the food contact substance list, forcing manufacturers to seek alternative preservatives for packaging linings and coatings.
FSMA Rule 204 and Traceability Requirements
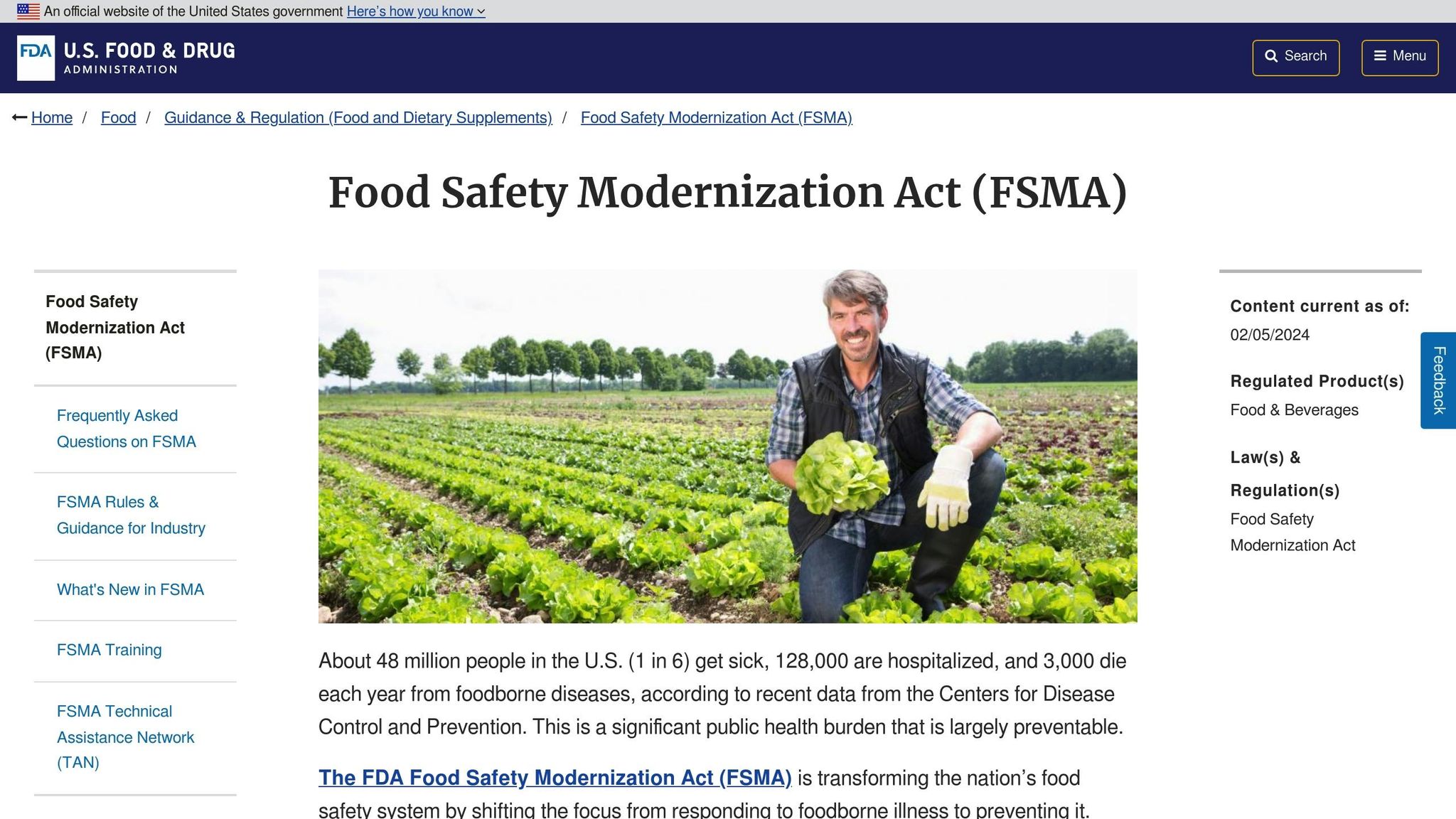
The compliance deadline for FSMA Rule 204, initially set for January 20, 2026, has been extended to July 20, 2028, following a Congressional directive. Despite the delay, the FDA plans to intensify its efforts in 2026 to educate and train businesses on traceability and recall procedures. For manufacturers using ingredients listed on the Food Traceability List - such as specific fish, nut butters, or fresh vegetables - enhanced traceability records are required. A "Transformation" Critical Tracking Event, which occurs when a food or its packaging undergoes changes, necessitates capturing detailed Key Data Elements linked to a Traceability Lot Code. Even with the extended deadline, companies are advised to continue developing their Traceability Plan, including protocols for recordkeeping and lot code assignment. These improvements align with the FDA's broader goal of enhancing packaging safety.
| Regulatory Initiative | 2026 Status | Impact on Packaging |
|---|---|---|
| GRAS Rulemaking | Proposed Rule (Spring 2026) | Ends self-affirmation; requires FDA notification for packaging substances |
| BHA Reassessment | Announced Feb 10, 2026 | May restrict common preservatives |
| FSMA Rule 204 | Enforcement Delayed to 2028 | Ongoing requirement to develop traceability plans and lot coding |
Compliance Best Practices for Pet Food Packaging
These practices are designed to align with FDA standards while safeguarding brand reputation by ensuring that all packaging components meet regulatory guidelines.
Material Verification and Supplier Transparency
Pet food packaging materials must adhere to the same FDA regulations as those for human food packaging. Manufacturers should obtain formal certifications from suppliers, confirming that all raw materials and finished packaging products comply with FDA food-contact standards. This ensures that each component is classified as Generally Recognized as Safe (GRAS), prior-sanctioned, or covered by an effective Food Contact Notification (FCN) or existing regulation under 21 CFR Parts 174 through 179.
The FDA provides a public database containing nearly 4,000 substances listed in its "Substances Added to Food" inventory. Regularly consulting the "GRAS Notice Inventory" and the "Effective Food Contact Substance Notifications" database can help verify that packaging materials meet legal standards. Additionally, all materials must meet "suitable purity" criteria for their intended use, though these standards may vary slightly from those applied to human food. These steps ensure that packaging materials comply with FDA regulations and support broader compliance efforts.
Avoiding Misleading Claims
To avoid misbranding, it’s essential to word claims carefully, adhering to the strict labeling rules discussed earlier. According to Section 201(m) of the FD&C Act, labeling includes all written, printed, or graphic materials on the container, wrapper, or accompanying items such as brochures. Claims suggesting disease treatment or altering body structure require approval through the new animal drug process.
For example, "Veterinarian Recommended" claims need to be backed by a statistically valid survey of veterinarians. On the other hand, terms like "Veterinarian Formulated" are permissible if at least one veterinarian has contributed to the formulation and this involvement is documented. However, "Veterinarian Approved" is not allowed, as only state regulatory agencies can approve products. Additionally, required label information must be at least 1/16 inch in height and displayed prominently, without being obscured by design elements (21 CFR 501.15).
GRAS Submission Processes: Old vs. New
The rules for GRAS submissions have shifted, moving from a voluntary system to a mandatory framework. This change strengthens pre-market oversight and reinforces the compliance principles outlined earlier. Previously, companies could self-determine GRAS status without notifying the FDA. Under the upcoming 2026 framework, all substances must be submitted for FDA review to ensure regulatory oversight and pre-market approval through the COSM module.
| Feature | Previous System (Self-Affirmation) | New 2026 Framework (Mandatory Submission) |
|---|---|---|
| Submission Requirement | Voluntary; companies could self-determine GRAS status without notifying FDA | Mandatory; substances must be submitted to the FDA for review |
| FDA Oversight | Limited; FDA reviewed voluntarily notified substances | Comprehensive; requires formal entry into the GRAS Notice Inventory |
| Transparency | Low; safety data often remained proprietary and internal to the company | High; safety data and FDA responses are maintained in a public database |
| Compliance Risk | Higher; self-determinations could be challenged by FDA after market entry | Lower; provides pre-market regulatory certainty through the COSM module |
"Because pet food is included in the general definition of food, a 'food additive' includes any substance the intended use of which results, or is reasonably expected to result, in its becoming a component of pet food or human food." - Pamela L. Langhorn, Author, PackagingLaw.com[2]
Conclusion
Meeting pet food packaging compliance means adhering to federal FDA standards while also keeping an eye on state-level regulations. The Food Safety Modernization Act (FSMA) has shifted the focus from reacting to contamination issues to proactively identifying potential hazards. This is achieved through written food safety plans and documented preventive controls. With the FDA-AAFCO Memorandum of Understanding expiring on October 1, 2024, the FDA now has sole responsibility for pre-market ingredient approvals and scientific reviews.
Pet food packaging must include the eight mandatory label elements required under federal labeling laws.
“As AAFCO's Pet Food Label Modernization standards are introduced, businesses should prepare for changes, such as human-style "Nutrition Facts" boxes and standardized handling instructions.” — Kevin Keating, President, PKG Brand Design
The recommended six-year transition period provides time for companies to adjust to these updates.
FAQs
Do I need an FDA Food Contact Notification (FCN) for my pet food packaging?
Pet food packaging generally doesn't require an FDA Food Contact Notification (FCN) unless the materials used are considered food contact substances that specifically need one. In most cases, pet food packaging adheres to the same FDA regulations as human food packaging to ensure safety and compliance.
How can I prove my packaging materials are FDA-compliant before launch?
To make sure your packaging materials align with FDA standards, it's crucial to verify that they meet the agency's regulations for food contact substances used in pet food packaging. These materials must be proven safe for food contact, which can be done through prior FDA approval, achieving GRAS (Generally Recognized as Safe) status, or conducting proper testing. Keeping thorough documentation of compliance and safety is a key part of the process.
While consulting with experts like PKG Brand Design can help communicate your compliance effectively, the ultimate responsibility for meeting FDA standards rests with manufacturers and suppliers.
What 2026 FDA changes should pet food brands plan for now?
Pet food brands need to gear up for new FDA regulations set to take effect on February 6, 2026. These updates are designed to make pet food packaging clearer and more consistent. Brands will be required to provide transparent ingredient lists, detailed nutritional information, and clear storage instructions on their labels.
This move builds on the July 2023 approval by AAFCO of updated model regulations, aimed at helping pet owners make more informed choices and fostering greater trust in pet food labeling.







